por
John R. Fischer, Senior Reporter | June 27, 2019
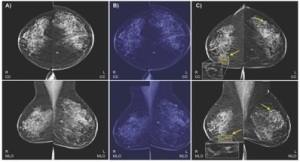
The algorithm is trained on EHR data
and mammogram images to predict
malignant breast cancer
A new AI model is just as good as human radiologists at predicting the development of malignant breast cancer in patients within the course of a year.
That’s the conclusion reached by a group of researchers at IBM Research – Haifa, who are responsible for developing an algorithm they say is the first to incorporate both mammogram and comprehensive electronic health record data. They hope to make the solution a “second reader” to aid radiologists in their analyses.
“Our algorithm takes into account many personal health factors, such as a family history of disease, breast density, and other elements that have been correlated with a higher risk of developing breast cancer,” Michal Rosen-Zvi, director of IBM Research, told HCB News. “It generates a per person risk prediction that is based on the unique combinations of many personal factors. Our hope is that this algorithm lays the groundwork for technology that could one day more accurately take into account all of these factors, and better assess a woman's risk from all of these predictive elements analyzed in conjunction with each other.”



Ad Statistics
Times Displayed: 107282
Times Visited: 6554 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
Potential types of findings that can be found in mammograms vary in shape, size, color and texture among other factors, making assessments challenging. While a second reading by another radiologist can increase sensitivity and specificity, the lack of trained radiologists and limited time often prevent providers in many countries from including second readers in standard screening procedures.
The aim of the system is to predict biopsy malignancy and differentiate normal from abnormal screening examinations, which it can do using both bilateral craniocaudal (CC) and mediolateral oblique (MLO) views, the typical processes used by radiologists.
The team developed and trained an algorithm with 9,611 clinically-collected, de-identified mammography images from 13,234 women who underwent at least one mammogram between 2013 and 2017 and had healthy records at least one year prior to their mammograms. The images were linked to holistic clinical data and biomarkers of each patient, such as thyroid function and reproduction history. Also incorporated were detailed records of each individual’s clinical data, including cancer history, pregnancy history, status of menopause, follow-up from biopsies, cancer registry data, lab results, and codes for various other procedures and diagnoses.
Utilizing deep learning and machine learning, the model maps connections between these and other additional clinical risk features to decrease the risk for breast cancer misdiagnosis and achieve accuracy comparable to radiologists, as defined by the American benchmark for screening digital mammography.

